|
Nature of Substituent Attached to the Atom: The carbon attached to more electronegative elements will possess a greater positive charge if a carbon atom is attached to more electronegative atoms while another is attached to less electronegative elements.ĭetermination of Electronegativity of an Element.The electronegativity also increases as the oxidation state of elements increases. This state of an element also influences its electronegativity. Oxidation state: It is the state that implies the number of electrons that an atom can lose, gain or share with another atom.For instance, if a carbon atom sp, sp 2, and sp 3 are hybridized in three compounds A, B, and C respectively, then, in these three compounds the order of electronegativity of carbon atom will be – Hybridization: The state of hybridization influences to a great extent the electronegativity of that element.When element A is slightly more electronegative than element B:įactors Affecting the Electronegativity of an Atomįollowing are the factors that affect the electronegativity of an atom:.When element B is slightly more electronegative than element A:.When the two atoms bonding together have equal electronegativity.Here's how they would look with their respective electronegative values: This will tell you how polar the bond is.Consider two atoms, A and B, with a tendency to attract bonding pairs of electrons, bond together. You look at the electronegativity of the two elements in the bond, and you calculate the difference. To find the electronegativity of a molecule, you need to find it for each bond. How do you find the Electronegativity of a molecule? It is not possible to measure electronegativity values for the noble gases, because they do not readily form bonds with other atoms. Do noble gases have an electronegativity? Fluorine is so electronegative, that it wants to rip an electron off anything it touches. This also explains the high reactivity of fluorine, chlorine and oxygen. Secondly, it means that oxygen always has a negative oxidation state, except in the very rare case where it forms a compound with fluorine. Firstly, it means that fluorine is always negative when combined with other elements. The Electronegativity of Chlorine, Fluorine and Oxygenįluorine is the most electronegative element on the electronegativity chart, followed by oxygen and then chlorine. And finally, if the difference is greater than 1.7 then the bond will be ionic. If the difference is between 0.4 and 1.7 then the bond is considered polar covalent. If the electronegativity difference between the two elements involved in bonding is less than 0.4 then the bond will be nonpolar covalent. Electronegativity Chart Why is electronegativity important?Įlectronegativity can tell us a lot about how different elements will bond to each other and which type of bond it will be. 
Halogens are considered to have a high electronegativity, while it is low for the alkali metals and alkaline earth metals. As a result, Fluorine is considered the most electronegative element while cesium is the least electronegative element. From top to bottom electronegativity decreases because of the increasing size of the atoms. This is because of the increased number of protons as the atomic number increase. Eletronegativity Trendsįrom left to right across the period table electronegativity increases. 
Attraction between protons and electrons means that atoms with a higher atomic number and number of protons have a higher electronegativity. This leads to larger atoms with more electron shells having lower electronegativity. The bigger the atom, the larger the distance, and the less effectively the protons are able to pull on the electrons. The positive protons in the nucleus “pull” on the negative electrons in the orbitals. Size is an important element of electronegativity. There are a variety of factors that affect the electronegativity of an atom. What affects the electronegativity of an atom? He did not come up with the concept, which was introduced by Jöns Jacob Berzelius in 1811, but he did develop a measurable scale, circa 1935. 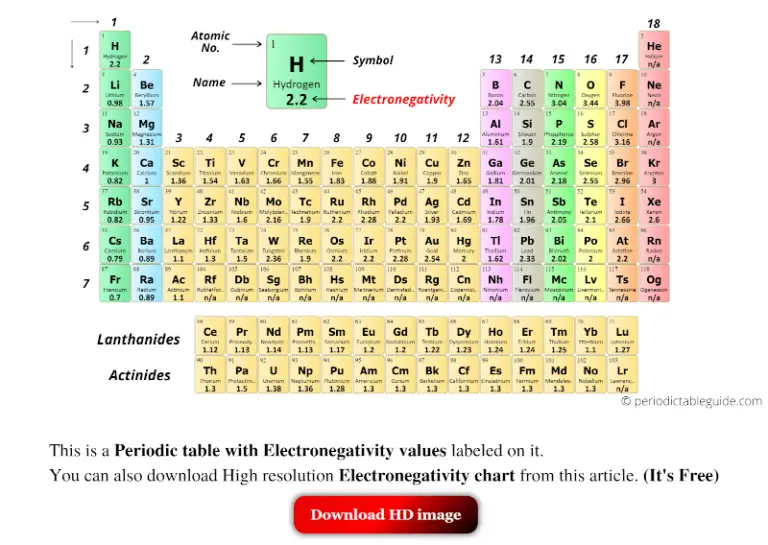
Pauling set the definition of electronegativity as “the power of an atom in a molecule to attract electrons to itself”. We will be exploring the electronegativity trends in the periodic table.Įlectronegativity was developed by Linus Pauling, who won the Noble prize twice. The higher the electronegativity, the stronger an atom attracts electrons. It is often viewed on an electronegativity chart of the elements, where trends and values can easily be seen. Different elements have different electronegativities based on a number of factors such as size and number of protons, neutrons, and electrons. There are several different ways of measuring it, the most common being the Pauling scale. Electronegativity is defined as an atom’s ability to attract electrons towards it in a chemical bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
